Themes & Goals
We aim to characterize the functional circuit-level processes of memory with which specific disorders can be understood, diagnosed, treated and cured.
Human memory is well specified by cognitive principles such as encoding, mnemonic search, recall, proactive interference, and retrieval induced forgetting. In contrast, the processing dynamics behind these operations take place at the circuit level and remain woefully unspecified. Pathologies of memory associated with Alzheimer’s Disease, post-traumatic stress disorder, schizophrenia, autism and normal aging place an enormous burden on individuals, relationships and societies. The development of treatments and cures for these pathologies require a functional understanding of the circuit-based mechanisms of memory.
Human memory is well specified by cognitive principles such as encoding, mnemonic search, recall, proactive interference, and retrieval induced forgetting. In contrast, the processing dynamics behind these operations take place at the circuit level and remain woefully unspecified. Pathologies of memory associated with Alzheimer’s Disease, post-traumatic stress disorder, schizophrenia, autism and normal aging place an enormous burden on individuals, relationships and societies. The development of treatments and cures for these pathologies require a functional understanding of the circuit-based mechanisms of memory.
How do neural circuits give rise to human memory? To answer this question, we combine optogenetics, DREADDs, pharmacology and behavioral manipulations with high-density tetrode and depth-probe recordings of neural activity in awake behaving rats. We are most interested in areas known as the hippocampus, medial septum, and entorhinal cortex which have all been shown to have important roles in memory in humans and animals. We use computational modeling to bridge this experimental work and human memory processing. This work suggests that neural rhythms allow the brain to code, manipulate and store information and that these dynamics are regulated by acetylcholine.
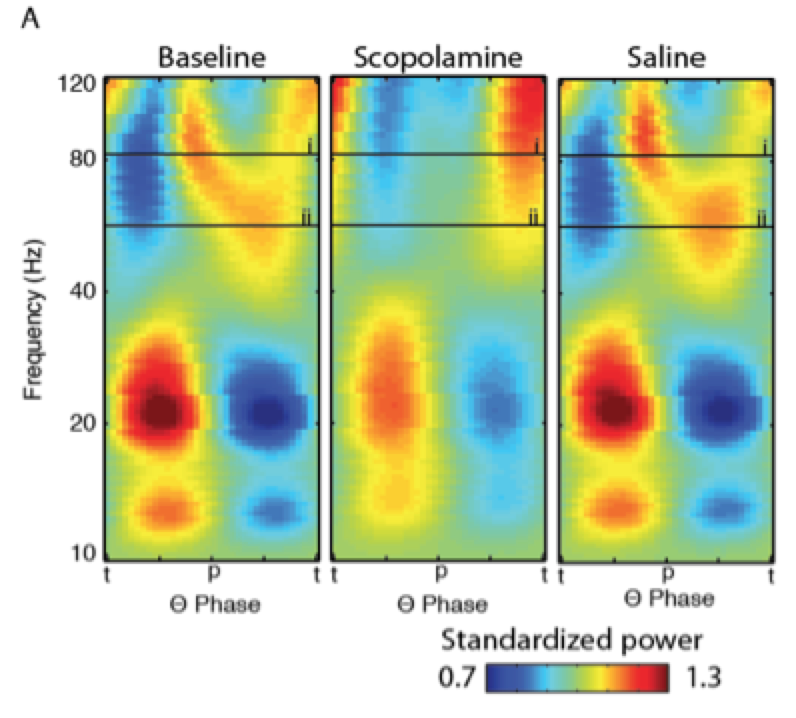
Systemic blockade of acetylcholine receptors results in a number of deficits in neural processing. These include the loss of key neural rhythms and spatial coding in the entorhinal cortex (Newman et al., 2013; 2014) and the loss of phase coding in hippocampal place cells (Newman et al., in prep.). These effects give rise to working hypotheses regarding the source of the cognitive deficits that accompany the loss of cholinergic signaling in the elderly or those with Alzheimer’s Disease. We will be following these hypotheses to 1) connect these markers to their respective cognitive deficits and 2) to dissect the relevant neurophysiology to discover the mechanisms of these effects with the goal of identifying avenues for future therapies or cures. We are currently following up these studies by optogenetically and chemogenetically manipulating the cholinergic system to determine the respective sites of action while recording neuronal activity to understand how these circuits are involved in memory.
More generally, this research aims to develop a systems-level understanding of how memories are encoded and retrieved. One outcome will be the development of a ‘functional lens’ through which psychological disorders can be understood and diagnosed, analogous to the microscope used by Alois Alzheimer to identify plaques and tangles in his famous patient. Toward this end, the lab seeks to develop new tools to observe and track neural activation.
More generally, this research aims to develop a systems-level understanding of how memories are encoded and retrieved. One outcome will be the development of a ‘functional lens’ through which psychological disorders can be understood and diagnosed, analogous to the microscope used by Alois Alzheimer to identify plaques and tangles in his famous patient. Toward this end, the lab seeks to develop new tools to observe and track neural activation.
Approaches